The Chen-Liu Laboratory
Investigate cancer as a biology and a treatment target
Principal Investigators
Chong Chen, PhD- PhD, University of Michigan-Ann Arbor (2009)
- BS, Peking University (1999)
| Yu Liu, PhD- PhD, Albert Einstein College of Medicine (2008)
- BS, Beijing Normal University (1997)
|
Research Interests
The Chen-Liu lab aims to understand the biology of cancer and identify new therapeutic targets for treatment. We develop and apply precision cancer models, genome editing, organoid culturing, multi-omics analyses, bioinformatics, and clinical studies to build a full technique chain from basic cancer studies to clinic oncology practice. We are proud of being a diverse and collaborating group of students, postdoc, physicians, staff, and faculty. Our current research focus on:
1. Precision cancer models
Taking advantage of recent progress in cancer genomics, genome editing, and organoid culturing, we develop a multiplexing strategy to create primary, orthotopic, and drivers-defined cancer models in mice. These precision cancer models can reproduce the whole process of tumorigenesis and recapitulate the pathology of human malignancies. So far, we have generated such precision animal models for lung cancer (NSCLC and SCLC), esophageal cancers (ESCC and EAC), gastric cancers, bladder cancer, nasopharyngeal cancer, and blood cancers (AML and lymphoma).
2. Tumor initiation and progression
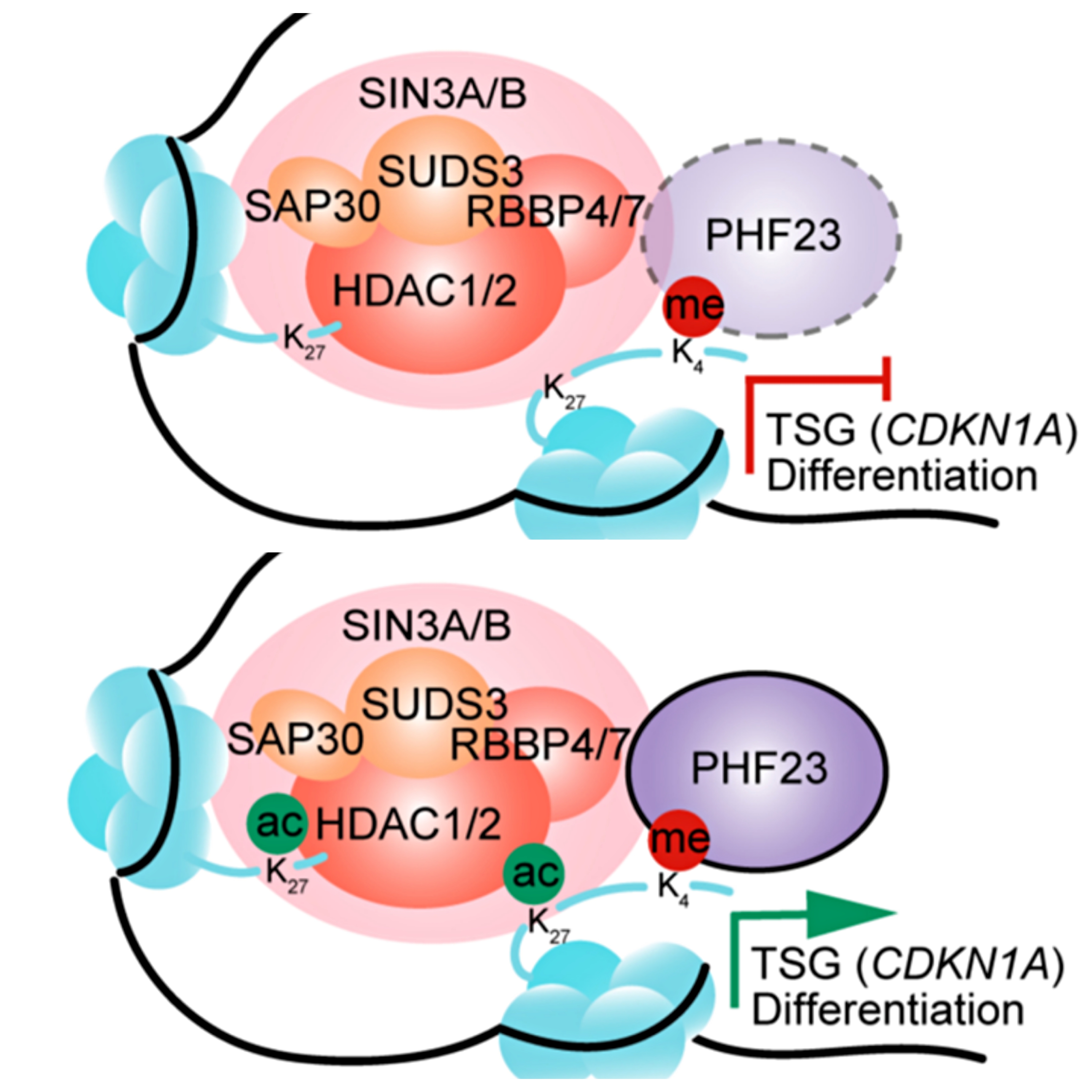
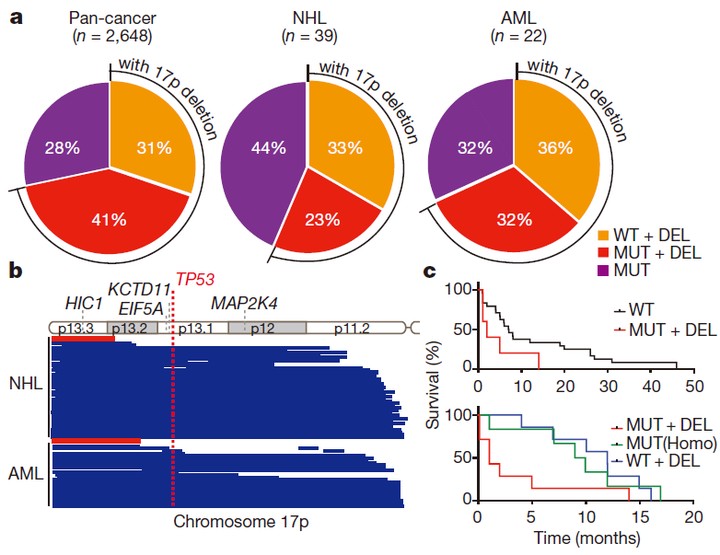
With these models, we investigate the detailed molecular mechanisms underlying tumorigenesis. We are especially interested in potential functional roles of human cancer-associated copy number variations (CNVs), a very much but less studied feature of human cancers. We have validated chromosome 17p loss as a driver for lymphoma and leukemia (nature 2016). We identify new tumor suppressors and oncogenes on CNVs, such as ALOX15B and PHF23 on chromosome 17p (nature 2016 and cancer discovery 2021) and KMT2C on chromosome 7q (cancer cell 2014). Interestingly, many of these genes are epigenetic factors. We explore the detailed molecular mechanisms of the epigenetic reprogramming in tumorigenesis.
3. Acquired chemo- and immunotherapy resistance
We apply our models and collaborate with physicians from the West China Hospital to dissect the molecular mechanisms underlying acquired resistance for cancer treatments. Ongoing projects includes studies on the acquired resistance to cisplatin-based chemotherapies in SCLC and MIBC and to immunotherapies in various types of cancers.
Selected publications
|
|
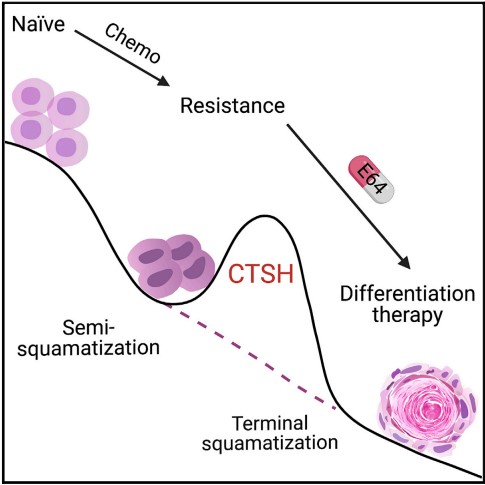
Acquired semi-squamatization during chemotherapy suggests differentiation as a therapeutic strategy for bladder cancer
Wang M, Chen X, Tan P, Wang Y, Pan X, Lin T, Jiang Y. et al. Cancer Cell (2022). https://doi.org/10.1016/j.ccell.2022.08.010
|
|
|
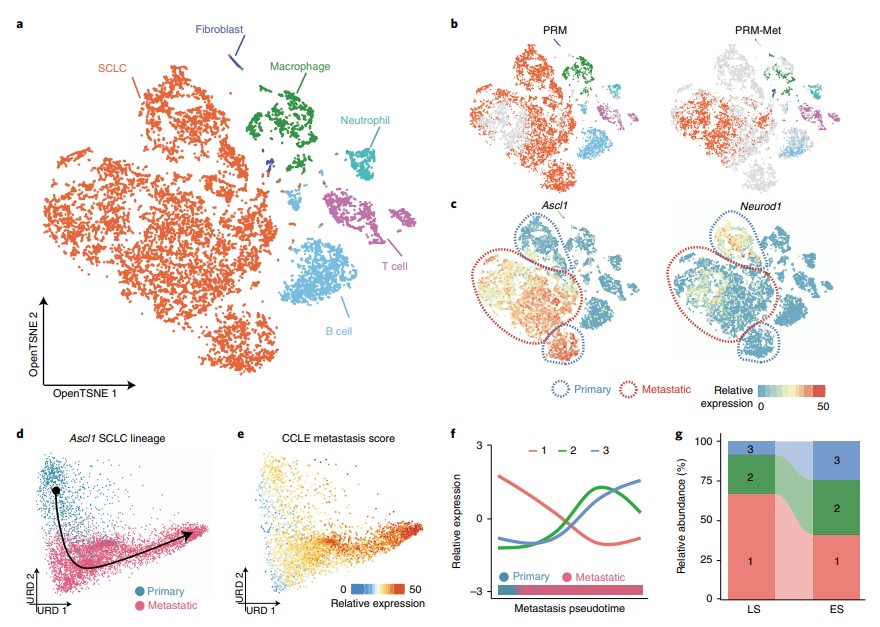
KMT2C deficiency promotes small cell lung cancer metastasis through DNMT3A-mediated epigenetic reprogramming
Na F, Pan X, Chen J, Chen X, Wang M, Chi P, You L. et al. Nature Cancer (2022). https://doi.org/10.1038/s43018-022-00361-6
|
|
|
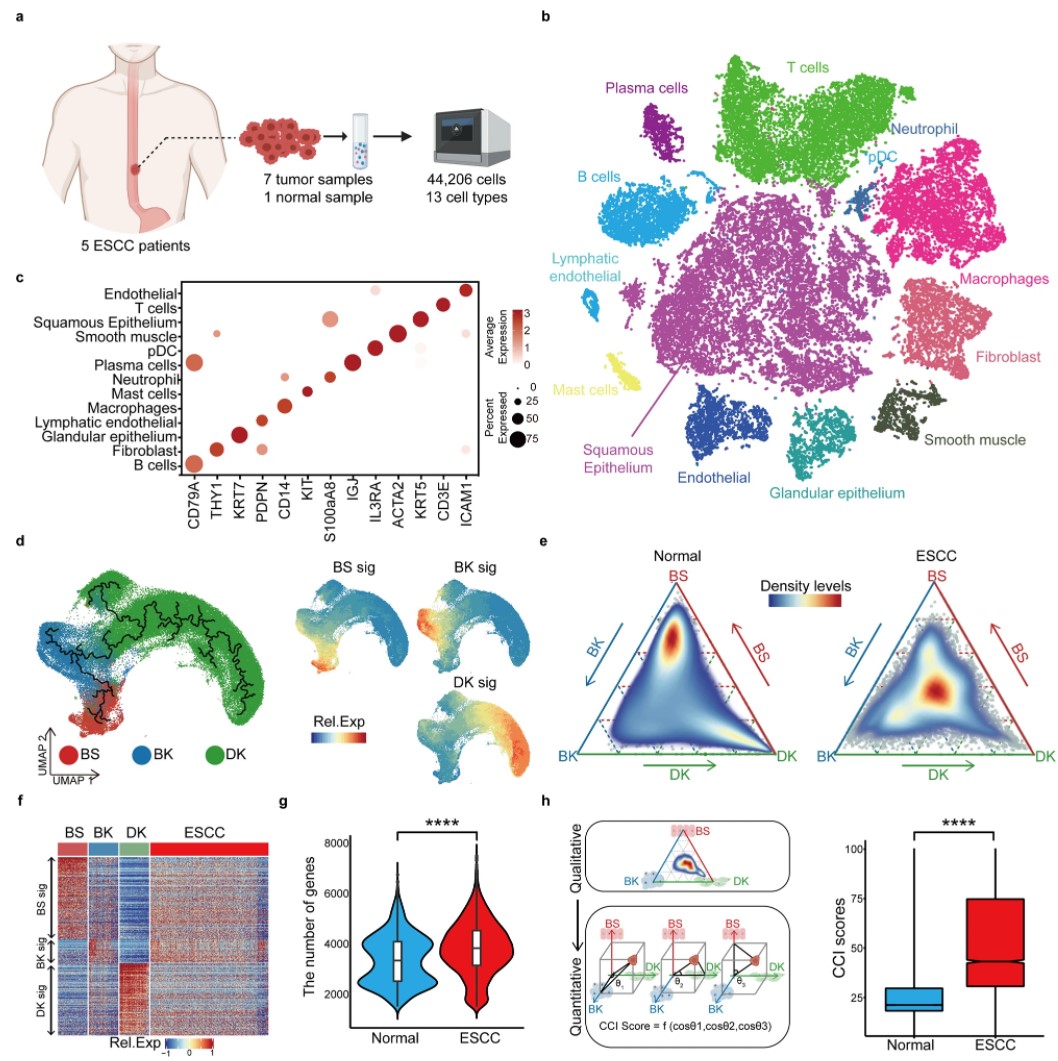
Identifying a confused cell identity for esophageal squamous cell carcinoma
Pan X, Wang J, Guo L, Na F, Du J, Chen X, Zhong A, Zhao L, Zhang L, Zhang M, Wan X, Wang M, Liu H, Dai S, Tan P, Chen J, Liu Y, Hu B, Chen C. Signal Transduct Target Ther. 2022 Apr 13;7(1):122. doi: 10.1038/s41392-022-00946-8. PMID: 35418165.
|
|
|
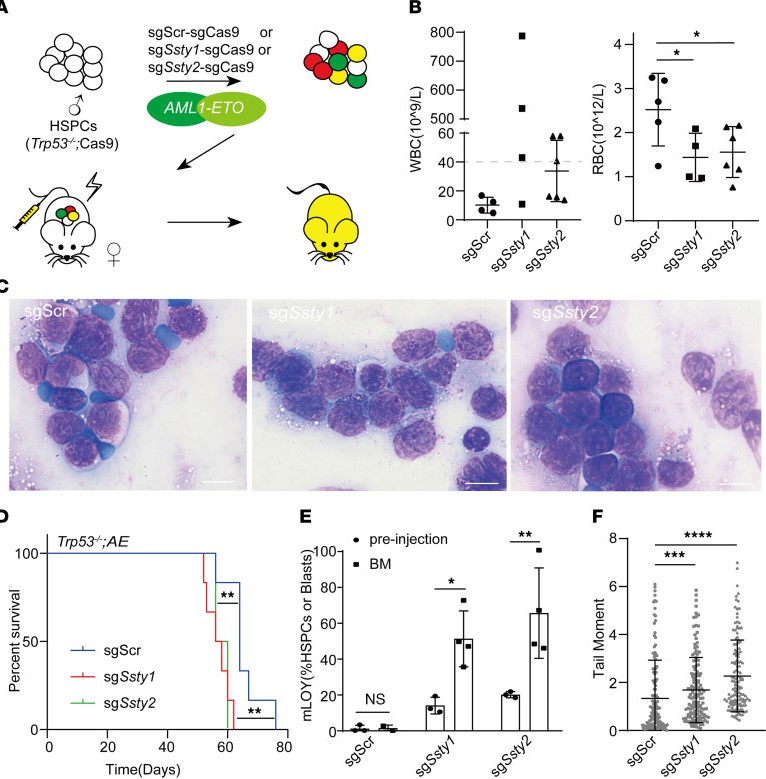
Mosaic loss of chromosome Y promotes leukemogenesis and clonal hematopoiesis
Zhang Q, Zhao L, Yang Y, Li S, Liu Y, Chen C. JCI Insight. 2022 Feb 8;7(3):e153768. doi: 10.1172/jci.insight.153768. PMID: 35132955; PMCID: PMC8855789.
|
|
|
An Epigenetic Mechanism Underlying Chromosome 17p Deletion-Driven Tumorigenesis
Chen M, Chen X, Li S, Pan X, Gong Y, Zheng J, Xu J, Zhao C, Zhang Q, Zhang S, Qi L, Wang Z, Shi K, Ding BS, Xue Z, Chen L, Yang S, Wang Y, Niu T, Dai L, Lowe SW, Chen C, Liu Y. Cancer Discov. 2021 Jan;11(1):194-207. doi: 10.1158/2159-8290.CD-20-0336. Epub 2020 Sep 25. PMID: 32978226; PMCID: PMC8591992.
|
|
|
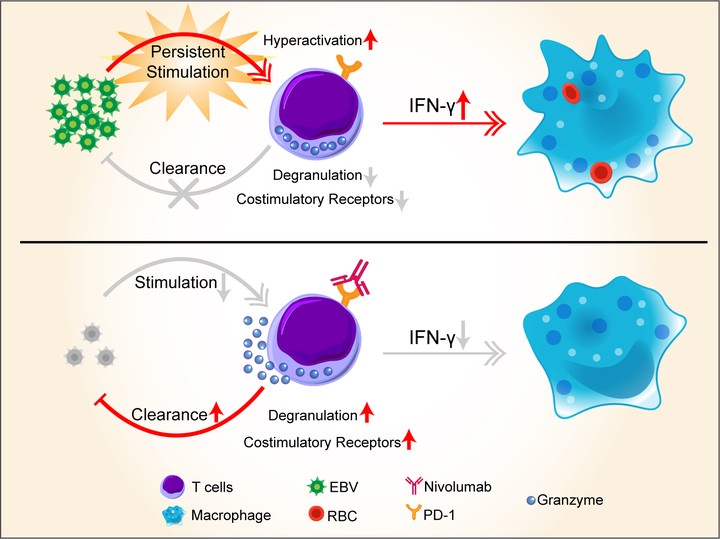
Nivolumab treatment of relapsed/refractory Epstein-Barr virus-associated hemophagocytic lymphohistiocytosis in adults
Liu P, Pan X, Chen C, Niu T, Shuai X, Wang J, Chen X, Liu J, Guo Y, Xie L, Wu Y, Liu Y, Liu. Blood. 2020 Mar 12;135(11):826-833. doi: 10.1182/blood.2019003886. PMID: 31914172.
|
|
|
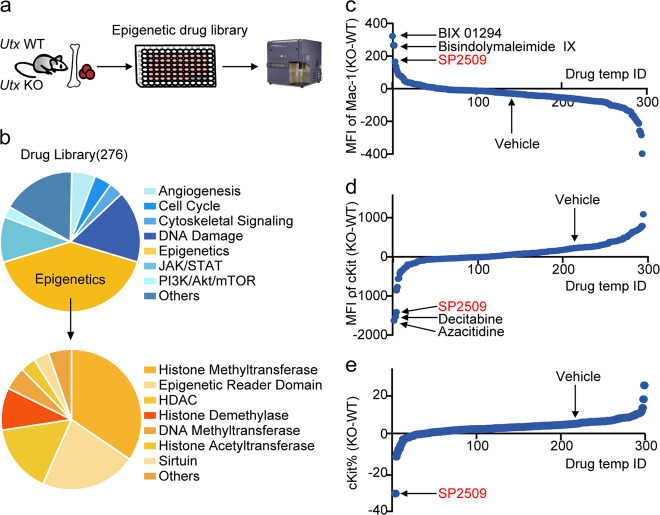
Epigenetic drug library screening identified an LSD1 inhibitor to target UTX-deficient cells for differentiation therapy
Wu B, Pan X, Chen X, Chen M, Shi K, Xu J, Zheng J, Niu T, Chen C, Shuai X, Liu Y. Signal Transduct Target Ther. 2019 Apr 26;4:11. doi: 10.1038/s41392-019-0040-2. PMID: 31044091; PMCID: PMC6483994.
|
|
|
Deletions linked to TP53 loss drive cancer through p53-independent mechanisms
Liu Y, Chen C, Xu Z, Scuoppo C, Rillahan CD, Gao J, Spitzer B, Bosbach B, Kastenhuber ER, Baslan T, Ackermann S, Cheng L, Wang Q, Niu T, Schultz N, Levine RL, Mills AA, Lowe SW. Nature. 2016 Mar 24;531(7595):471-475. doi: 10.1038/nature17157. Epub 2016 Mar 16. PMID: 26982726; PMCID: PMC4836395.
|